|
As early as 1869, Russian chemist Dimitri Mendeleev began arranging the then-known elements by atomic weight, creating what we now call the. By 1809, at least 47 were known, and by the mid-1800’s, naturalists were attempting to organize them. Click on 'Element Atomic Number', 'Element Symbol', 'Element Name' and 'Element Color' headers to sort. As early as 1669, naturalists began to discover individual elements. Although rather advanced for school pupils, their teachers will find it useful.This Color table gives the Color of all the elements of periodic table. Aimed at chemistry teachers and students at all levels, it advises on the best presentation of formulae and chemical graphics. The book is replete with examples for guidance and there are extensive tables to direct the reader to information quickly. 'If we move from red all the way up to violet, the frequency. demonstrating on a toy color-coded xylophone. New developments are described and additional nomenclature systems used in specific industries are introduced. Were all familiar with the elements of the periodic table. The book clearly explains the fundamental principles of nomenclature methods and enables the reader to apply them accurately and with confidence. It also includes some basic biochemical nomenclature. Follow the steps in this video to color code your periodic table to show groups of metals, non-metals, and metalloids solids, liquids, and gases and the pe. This periodic table is suitable for a computer and mobile device wallpaper. If you like the design or display of this periodic table rather than that of the others, you can print it. The p-block is a mix of metals, metalloids, and non-metals as well. Visualize trends, 3D orbitals, isotopes, and mix compounds. The periodic table contains four blocks: s-block, p-block, d-block, and f-block. It is also available in high resolution pdf. Interactive periodic table showing names, electrons, and oxidation states. Alternately, you can also use the printable one. The IUPAC doesnt update these values annually, so these are the most recent values for 2019. When you click it, it will show you the detail information about the element. 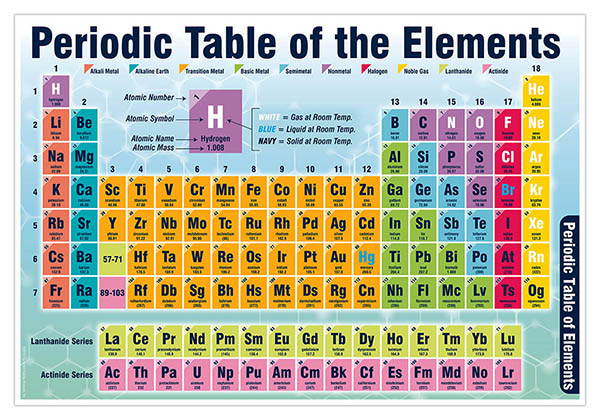
Written by leading world authorities this second edition of Principles of Chemical Nomenclature outlines IUPAC recommendations for application in the principle branches of chemistry: organic, inorganic, organometallic, and polymer. This color periodic table contains the accepted standard atomic weights (atomic masses) of each element as accepted by the IUPAC. 
This requires a rational basis from which comprehensible names can be developed. The ultimate aim of IUPAC nomenclature is to provide unequivocal and comprehensible names for all kinds of chemical structures. The IUPAC doesn't update these values annually, so these are the most recent values for 2019. /Periodic-Table-Color-58b5c80f5f9b586046cae1a6.png)
The various blocks of elements are colour-coded: s-block, yellow p-block, green d-block, blue f-block, orange. This color periodic table contains the accepted standard atomic weights (atomic masses) of each element as accepted by the IUPAC. This is because the isotope ratio greatly depends on the element sample source. This colored periodic table is a brightly colored periodic table suitable for printing. You may notice the table includes a range of values for most elements. Place black dots in the squares of all alkali metals. Using map pencils, color each group on the table as follows: Color the square for Hydrogen sky blue. Your teacher will give you a copy of the periodic table to color. However, many students struggle to understand the periodic table due to its sheer complexity. This worksheet will help you understand how the periodic table is arranged. Its a complex system of elements thats used to classify and study substances. Principles of Chemical Nomenclature: A Guide to IUPAC Recommendations 2011 Edition, edited by Jeff Leigh, RSC Publishing, 2011 Recommended values from Table 1 for the atomic polarizabilities (atomic units estimated uncertainties in parentheses) of elements Z 1120. In December, 2018, the IUPAC updated its periodic table to include revisions to the atomic weight values. Table 51 sets out the specific designation system of colour or alphanumeric code for identification conductors, and is referenced by Regulations 514.4.4, 514.4.6 and 514.5.3. In the world of chemistry, the periodic table is a cornerstone of scientific discovery.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
